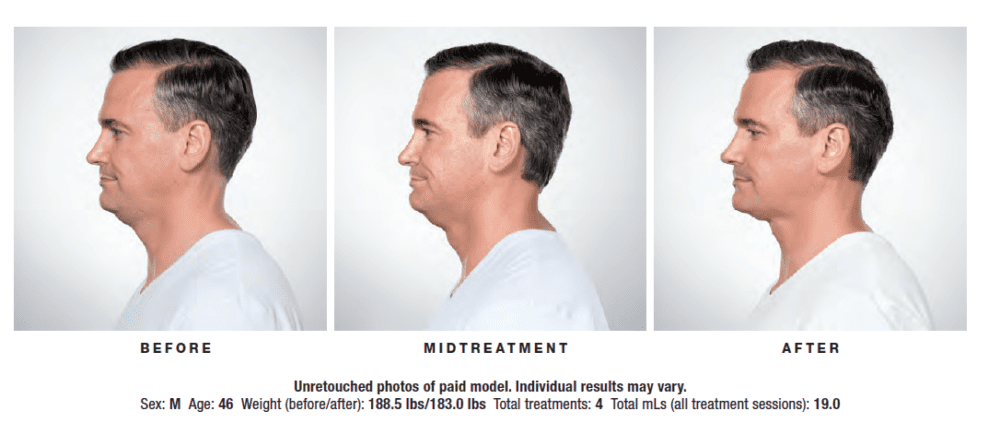
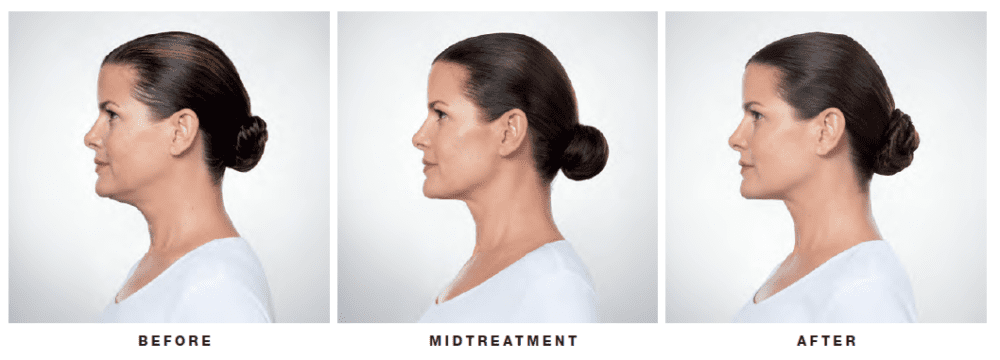
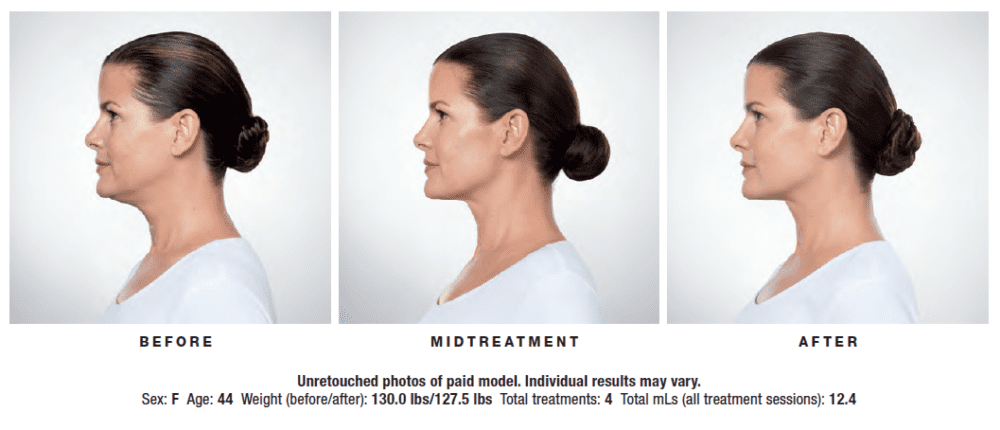
KYBELLA® is a prescription medicine and is the only FDA-approved injectable treatment used in adults to improve the appearance and profile of moderate to severe fat below the chin (submental fat), also called “double chin.” Submental fullness is a gathering of excess fat beneath the chin, commonly known as “double chin.” Once treated, the fat cells are destroyed and leave your body through natural processes.
Your ClearlyDerm provider will create a tailored treatment plan based on your chin profile. At each treatment, you will be given multiple small injections under your chin. The number of injections required depends on the size of the treatment area and your desired results. You may receive up to 6 treatment sessions, spaced at least 1 month apart. Many patients experience visible results in 2 to 4 KYBELLA® treatments. 59% of patients received 6 treatments in clinical studies.
Kybella is commonly used to treat:
- The appearance and profile of moderate to severe fat below the chin (submental fat), also called “double chin
What To Expect
- Length Of Time For The Procedure: 15-30 minutes depending on the size and area being treated. Up to 6 treatments, one month apart.
- Recovery Time: There is little to no downtime following the procedure. Some patients can experience slight bruising, swelling, numbness and redness, which typically subsides soon after the procedure.
- Final Results Of The Procedure: 12-24 weeks depending on the amount of secessions
- Duration of Kybella: fat cells are permanently removed