
Pemphigus Vulgaris Clinical Trial
Pemphigus Vulgaris Clinical Trial
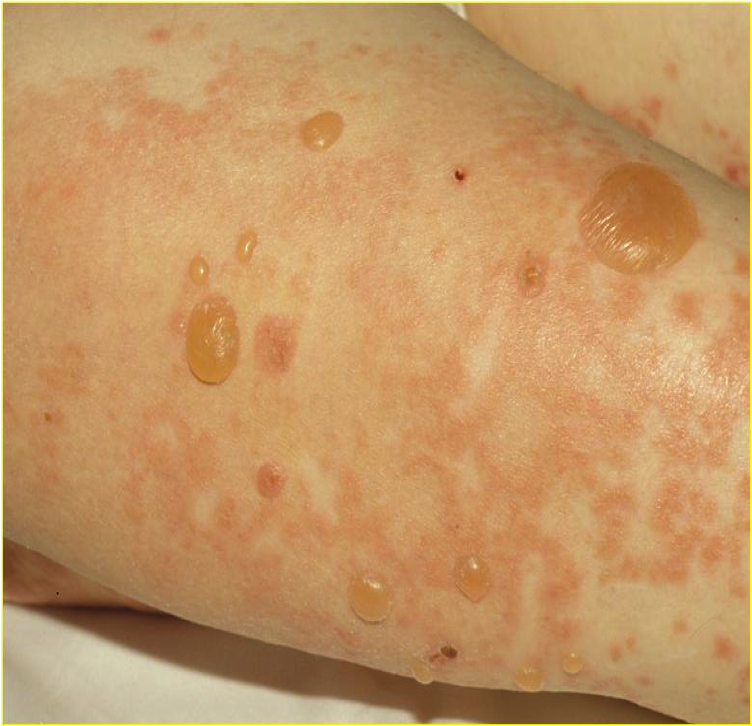
Patients with moderate to severe Pemphigus Vulgaris, also known as a family of autoimmune diseases that affect the skin and mucous membranes and cause blisters and sores in the mouth and throughout the body, will be enrolled to evaluate new treatments for this skin condition. Clinical trial participants will be monitored closely by medical professionals for the duration of the study. The goal of this Pemphigus Vulgaris clinical trial is to develop better understanding of Pemphigus Vulgaris and develop safe and effective therapies.
Study Snapshot
Duration
Up to 136 weeks depending on study
Incentive
Subject compensation and travel assistance available. Study medication, study related exams, and lab tests will be provided at no cost.
Have questions about this clinical trial?
For general questions regarding our clinical trials, please contact our team at clinicaltrials@clearlyderm.com or fill out the contact form below.
Frequently Asked Questions
Click or Press on the Questions to See the Answers.
A clinical trial is a research study conducted with human volunteers to test the effectiveness and safety of new drugs, devices, or interventions. Our Clearlyderm clinical trials focus on skin conditions such as psoriasis, acne, rosacea, or atopic dermatitis (eczema).
To enroll in one of our clinical trials, contact our office. You will need to review and complete informed consent documents and complete screening procedures prior to participation.
Like any medical treatment, potential risks exist. However, participants are informed of these risks prior to starting the clinical trials and receive monitoring and follow-up care throughout the trial duration if selected to participate.
Pemphigus vulgaris (PV) is a rare autoimmune disease. This skin disorder causes blisters on the skin and mucous membranes. The blisters itch and can burst easily. While this disease cannot be cured, treatment can reduce, and even clear the blisters and sores. PV affects the eyes, nose, mouth, throat and genitals.
Schedule an appointment
Looking to visit a dermatologist? Click the button below to schedule an appointment with our team of skin experts.




